Wouldn’t it be great if we had a way to tell genetic variants that do something to gene function and regulation from those that don’t? This is a Really Hard Problem, especially for variants that fall outside of protein-coding regions, and thus may or may not do something to gene regulation.
There is a host of bioinformatic methods to tackle the problem, and they use different combinations of evolutionary analysis (looking at how often the position of the variant differs between or within species) and functional genomics (what histone modifications, chromatin accessibility etc are like at the location of the variant) and statistics (comparing known functional variants to other variants).
When a new method is published, it’s always accompanied by a receiver operating curve showing it predicting held-out data well, and some combination of comparisons to other methods and analyses of other datasets of known or presumed functional variants. However, one wonders how these methods will do when we use them to evaluate unknown variants in the lab, or eventually in the clinic.
This is what this paper, Liu et al (2019) Biological relevance of computationally predicted pathogenicity of noncoding variants is trying to do. They construct three test cases that are supposed to be more realistic (pessimistic) test beds for six noncoding variant effect predictors.
The tasks are:
- Find out which allele of a variant is the deleterious one. The presumed deleterious test alleles here are ones that don’t occur in any species of a large multiple genome alignment.
- Find a causative variant among a set of linked variants. The test alleles are causative variants from the Human Gene Mutation Database and some variants close to them.
- Enrich for causative variants among increasingly bigger sets of non-functional variants.
In summary, the methods don’t do too well. The authors think that they have ‘underwhelming performance’. That isn’t happy news, but I don’t think it’s such a surprise. Noncoding variant prediction is universally acknowledged to be tricky. In particular, looking at Task 3, the predictors are bound to look much less impressive in the face of class imbalance than in those receiver operating curves. Then again, class imbalance is going to be a fact when we go out to apply these methods to our long lists of candidate variants.
Task 1 isn’t that well suited to the tools, and the way it’s presented is a bit silly. After describing how they compiled their evolution-based test variant set, the authors write:
Our expectation was that a pathogenic allele would receive a significantly higher impact score (as defined for each of the six tested methods) than a non-pathogenic allele at the same position. Instead, we found that these methods were unsuccessful at this task. In fact, four of them (LINSIGHT, EIGEN, GWAVA, and CATO) reported identical scores for all alternative alleles at every position as they were not designed for allelic contrasts …
Sure, it’s hard to solve this problem with a program that only produces one score per site, but you knew that when you started writing this paragraph, didn’t you?
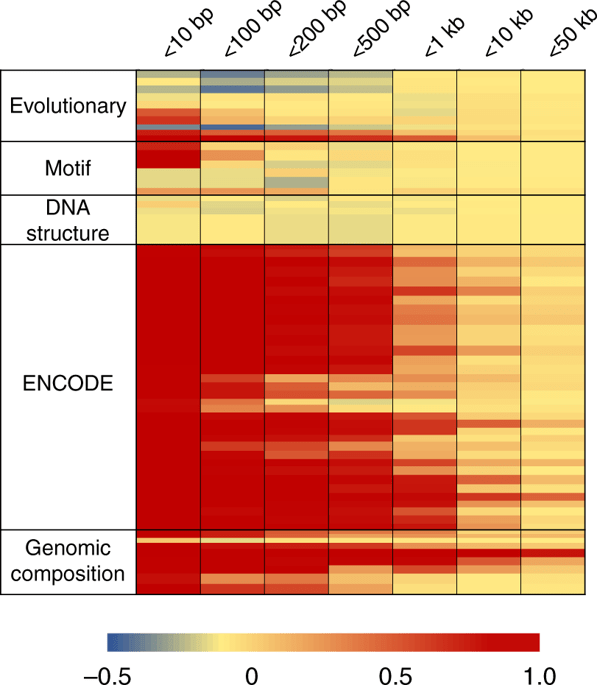
The whole paper is useful, but to me, the most interesting insight is that variants close to each other tend to have correlated features, meaning that there is little power to tell them apart (Task 2). This might be obvious if you think about it (e.g., if two variants fall in the same enhancer, how different can their chromatin state and histone modifications really be?), but I guess I haven’t thought that hard about it before. This high correlation is unfortunate, because that means that methods for finding causative variants (association and variant effect prediction) have poor spatial resolution. We might need something else to solve the fine mapping problem.
Figure 4 from Liu et al., showing correlation between features of linked variants.
Finally, shout-out to Reviewer 1 whose comment gave rise to these sentences:
An alternative approach is to develop a composite score that may improve upon individual methods. We examined one such method, namely PRVCS, which unfortunately had poor performance (Supplementary Figure 11).
I thought this read like something prompted by an eager beaver reviewer, and thanks to Nature Communications open review policy, we can confirm my suspicions. So don’t say that open review is useless.
Comment R1.d. Line 85: It would be interesting to see if a combination of the examined scores would better distinguish between pathogenic and non-pathogenic non-coding regions. Although we suspect there to be high correlation between features this will test the hypothesis that each score may not be sufficient on its own to make any distinction between pathogenic and non-pathogenic ncSNVs. However, a combined model might provide more discriminating power than individual scores, suggesting that each score captures part of the underlying information with regards to a region’s pathogenicity propensity.
Literature
Liu, L., Sanderford, M. D., Patel, R., Chandrashekar, P., Gibson, G., & Kumar, S. (2019). Biological relevance of computationally predicted pathogenicity of noncoding variants. Nature Communications, 10(1), 330.